By Pamela Dozois
news@santaynezvalleystar.com
SCI Research Advancement continues to make important progress towards reaching the goal of finding a cure for paralysis. In yet another milestone for the local nonprofit, SCI Research Advancement was asked by the Santa Barbara Cottage Hospital Institutional Review Board to submit a request to be published in the National Clinical Trials.gov registry.
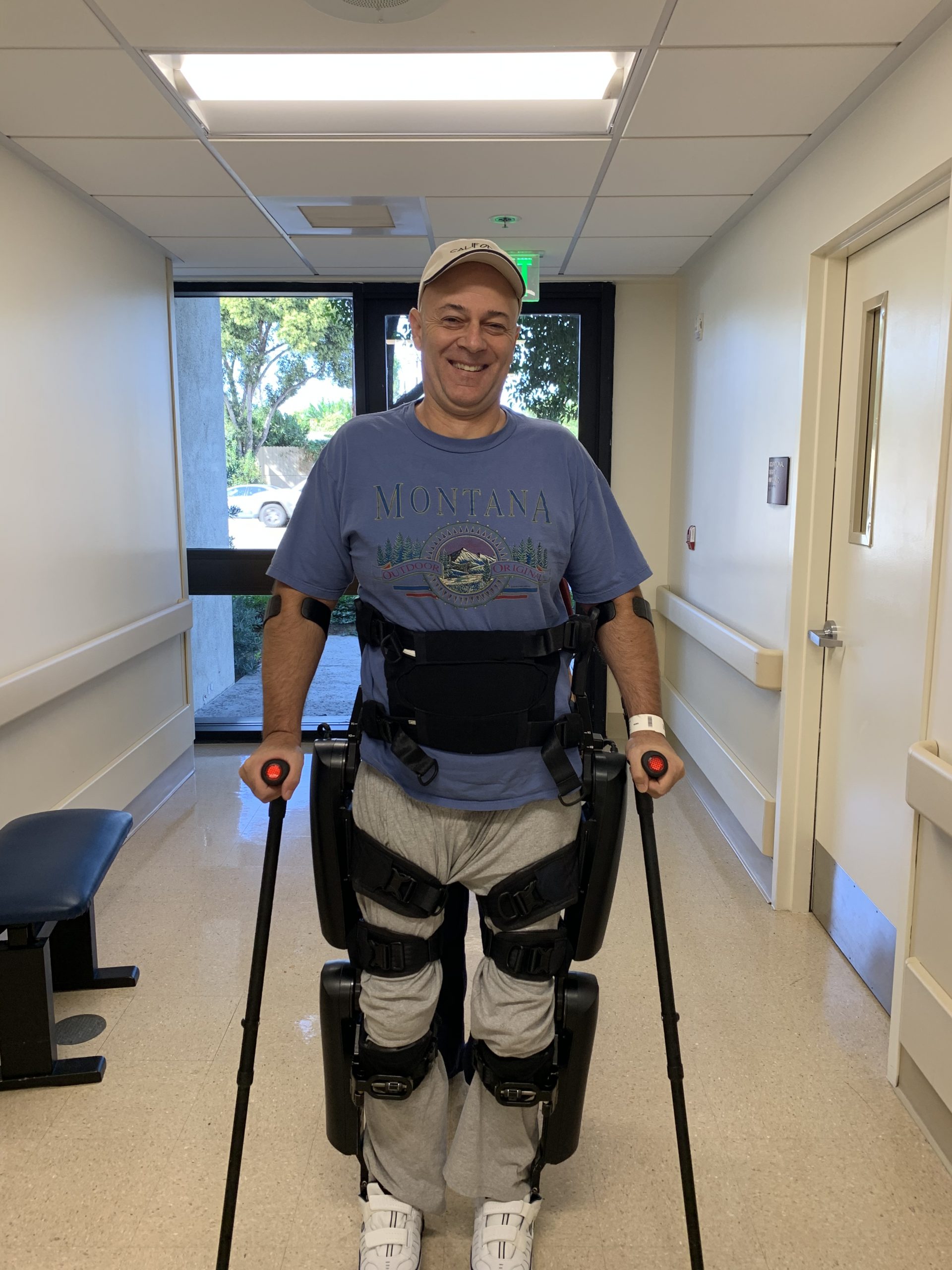
Last week, the ongoing Phase One study for spinal cord injury was approved for publication on the registry. The official case study can be seen on the ClinicalTrials.gov web site, Identifier Number is NCT04097015. Phase Two will be the combination of low-level electrical stimulation along with an exoskeleton for local motor training and therapeutic walking to stimulate dormant nerves and restore function.
“This is a significant achievement that will allow our medical data to be scrutinized, analyzed, and reviewed by anyone with an interest in medical research,” said Will Ambler, founder of SCI Research Advancement.
“Our intention is to add complementary medical procedures along with novel physical therapy to restore function that is lost after being paralyzed with spinal cord damage,” he explained. “Once our project is completed, we will then have a formal and medically legitimate procedure that can be expanded and later apply for FDA approval. This is a first but very important step in obtaining medical approval.”
The research team for the Phase One and Two of the clinical trial are Dr. Wendy Strouse-Watt, the principal investigator for the non-invasive electrical stimulation (NI-ES), and Dr. George O’Clock Ph.D., a researcher and published expert in low level electrical stimulation, who will supervise and advise the research project. Ambler will be contributor as well as first patient to provide feedback and evaluate efficacy of treatments.
“To begin the Second Phase of our study we are currently raising funds to purchase an exoskeleton,” Ambler said. “An exoskeleton will be a necessary and very important portion of the overall treatment.”
For more information, call 805-688-7755 or to make a donation via the Go Fund Me page, visit www.gofundme.com/stand-to-end-paralysis or a direct donation at the home page at www.scicure.org